
2016 Michael Sweeney Award Winners
The Michael A. Sweeney Endowed Award in Chemistry and Biochemistry was established with gifts from Dr. Sweeney’s children, as well as devoted colleagues, former students and friends to honor and remember Dr. Michael Sweeney, Professor Emeritus of Chemistry at Santa Clara University, where he taught for 46 years.
All Chemistry and Biochemistry majors are eligible to apply for this coveted award. Winners are selected by a committee consisting of permanent faculty members of the department. Winning entries this year came from Kyle Bandaccari ’16 and Daniel Tiano ’17. The committee judged that both Kyle and Daniel best exemply Dr. Sweeney’s passion for teaching, curiosity and humor as evidenced by the letters provided by their mentors and by the similes they created in the spirit of “Sweeney’s Similes.”
Kyle’s simile:
On chlorofluorocarbons (CFCs) in the atmosphere:
(At the risk of crossing into unabashed directness, parenthetical comments explain the actual chemistry I intend to reference)
The radical reactions catalyzed by CFCs which deplete the ozone layer are a lot like the times when you say something stupid in front of a group of friends. A seemingly innocuous observation (incident photon) suddenly makes one or two of them crack up laughing and makes you the center of attention (initiation via chlorine or fluorine radical dissociation). When you try to explain yourself to others nearby who didn’t hear it, the laughing becomes contagious and the entire situation blows up (propagation). Things only calm down when you find someone just as clueless who you can sympathize with and who already said something dumb just a few minutes earlier (recombination/termination). The good news is that you only have to watch everything you say and do for the next fifty or so years to restore your dignity (reduce CFC use/emissions during wait time for ozone layer repair).
Dan’s simile:
Atomic orbitals can be thought of as bedrooms within a house with the following floor plan:
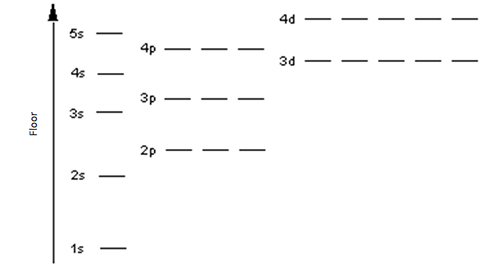
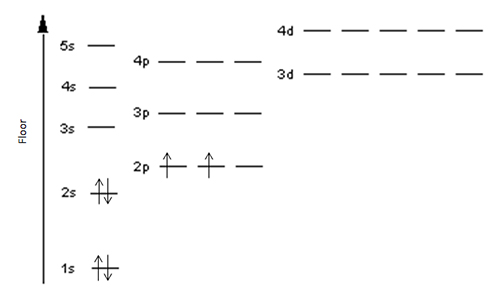
As you can see, each floor has one, three, or five bedrooms. Each is quite small and suitable for a maximum of two people, just as an orbital can hold a maximum of two electrons. Now, imagine having to pick your bedroom in this house. I don’t know about you, but I don’t want to have to climb several stories just to get to my bedroom, as it would take too much energy. Therefore, I would naturally pick the room on floor 1s. If 1s happened to be already filled up, I would go to the next easiest floor which would be 2s. After 2s would be 2p, then 3s, and so on. This trend would remain consistent no matter how many floors/rooms are occupied; just like an electron, I choose to occupy the room that takes the least amount of energy to get to.
For example, how does the Carbon family of six fill up their house? The bedrooms on floors 1s and 2s are occupied by mom, dad, grandma, and her weird new boyfriend, Eugene. This leaves the entire floor 2p for just the twin children. It wouldn’t make much sense to put them in the same bedroom since there are empty ones just down the hall, so they each get their own room. Bigger families, such as the Chlorine family and Silicon family will occupy more bedrooms on higher floors.
Dan Tiano; Michael Carrasco, Dept Chair; Kyle Bandaccari