Arts and Sciences Collaborates with Engineering in Nanomaterial Research
Nanomaterials represent a whole new world of possibility across a range of scientific studies, including medicine and engineering. But how exactly do these tiny particles, whether natural or man-made, interact with their environment?
That’s the question a cross-disciplinary group of SCU researchers set out to answer. In a study published this January in Environmental Science: Nano, Professors Korin E. Wheeler (Chemistry and Biochemistry) and Maryam Mobed-Miremadi (Bioengineering), in collaboration with recent Santa Clara graduates Matthew Findlay (Bioengineering ’17) and Daniel Freitas (Bioengineering ’17), described a predictive tool they had developed to describe the proteins that coat nanomaterials in order to apply nanomaterial technology in medicine, both for treatment and diagnosis. With strong reviewer comments, the article was selected for the journal’s cover.
Nanomaterials are “nano-sized [one millionth of a millimeter] particles” and can take the form of a variety of natural materials “such as carbon or minerals like silver,” according to the National Institute of Environmental Health Sciences. While nanomaterials have been under study for a while now, it has been difficult for scientists to quickly and accurately predict how nanomaterials will interact with their environment. “Their small size and surprising properties make them difficult to define, discuss, and evaluate,” said Dr. Wheeler.
“I think the best way to describe [the problem this project seeks to address] in layman’s terms is that we have these materials that are being used a lot by engineers because they have a lot of desirable properties that can help us with products,” Findlay explained. “The thing is, these materials enter the environment and we’re not exactly sure how they interact in the environment and how toxic they are.”
Professors Mobed-Miremadi and Wheeler and students Findlay and Freitas were initially brought together through the two students’ coursework in the College of Arts and Sciences. Freitas began researching with Professor Wheeler in the spring of his first year at Santa Clara. Findlay and Freitas became friends outside of class, and Findlay began working with Freitas and Wheeler. Mobed-Miremadi was asked to join the team after the two undergraduates realized that her contributions through her area of expertise, chemical engineering, would be essential to the success of the project. The data was generated in Professor Wheeler's lab and analyzed remotely.
Soon, the four researchers formed a cross-disciplinary dream team.
The group’s work moves forward in the process of perfecting how scientists can predict the interactions between nanomaterials and the proteins that coat them when they enter the body, a process that often renders the nanomaterials unrecognizable by the body. “Silver nanomaterials, for example, can dissolve to form silver ions in solution. Silver ions are considered nontoxic to humans at reasonable concentrations but can affect the growth of fish embryos,” Professor Wheeler explained in an article for Slate. “The formation of ionic silver is not only dependent on the engineered properties of the nanomaterial (like size or shape); it’s also dependent on changes from environmental exposure (for instance, coatings of biomolecules or surface imperfections).”
Mobed-Miremadi attributes their success to the fact that “Everyone knew exactly what they were doing.” She added “The effectiveness of the team stemmed from the fusion [of the team] and the strength of interdisciplinary collaboration.”
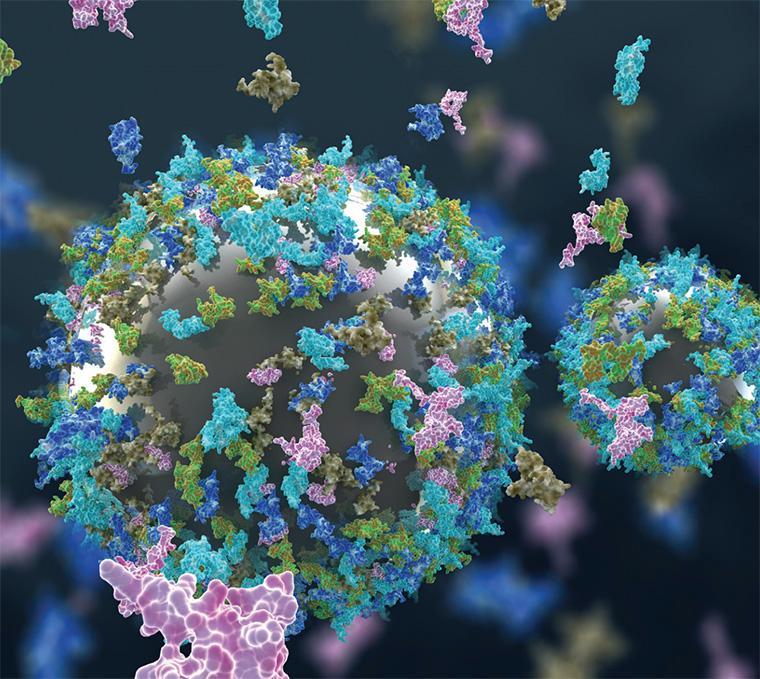
The illustration for the SCU research team’s cover article in Environmental Science Nano depicts nanomaterials interacting with proteins. Image courtesy of Ella Maru Studios